
Version Control Challenges in DMS Software and How to Solve Them
Learn the common version control challenges in DMS software and discover effective solutions to ensure document accuracy, compliance, collaboration, and audit readiness.
Expert perspectives on pharma manufacturing, compliance, AI automation, and digital transformation in life sciences.

Learn the common version control challenges in DMS software and discover effective solutions to ensure document accuracy, compliance, collaboration, and audit readiness.

Explore the biggest challenges pharmaceutical companies face during eBMR implementation, from process standardization and system integration to compliance and user adoption. Learn how modern eBMR software helps streamline batch manufacturing, improve data integrity, and accelerate digital transformation in pharma operations.

Switching from paper logbooks to electronic systems is helping GMP facilities improve compliance, reduce documentation errors, and increase operational efficiency. Discover how electronic logbooks support data integrity, faster audits, improved batch release processes, and measurable ROI in modern pharmaceutical manufacturing.
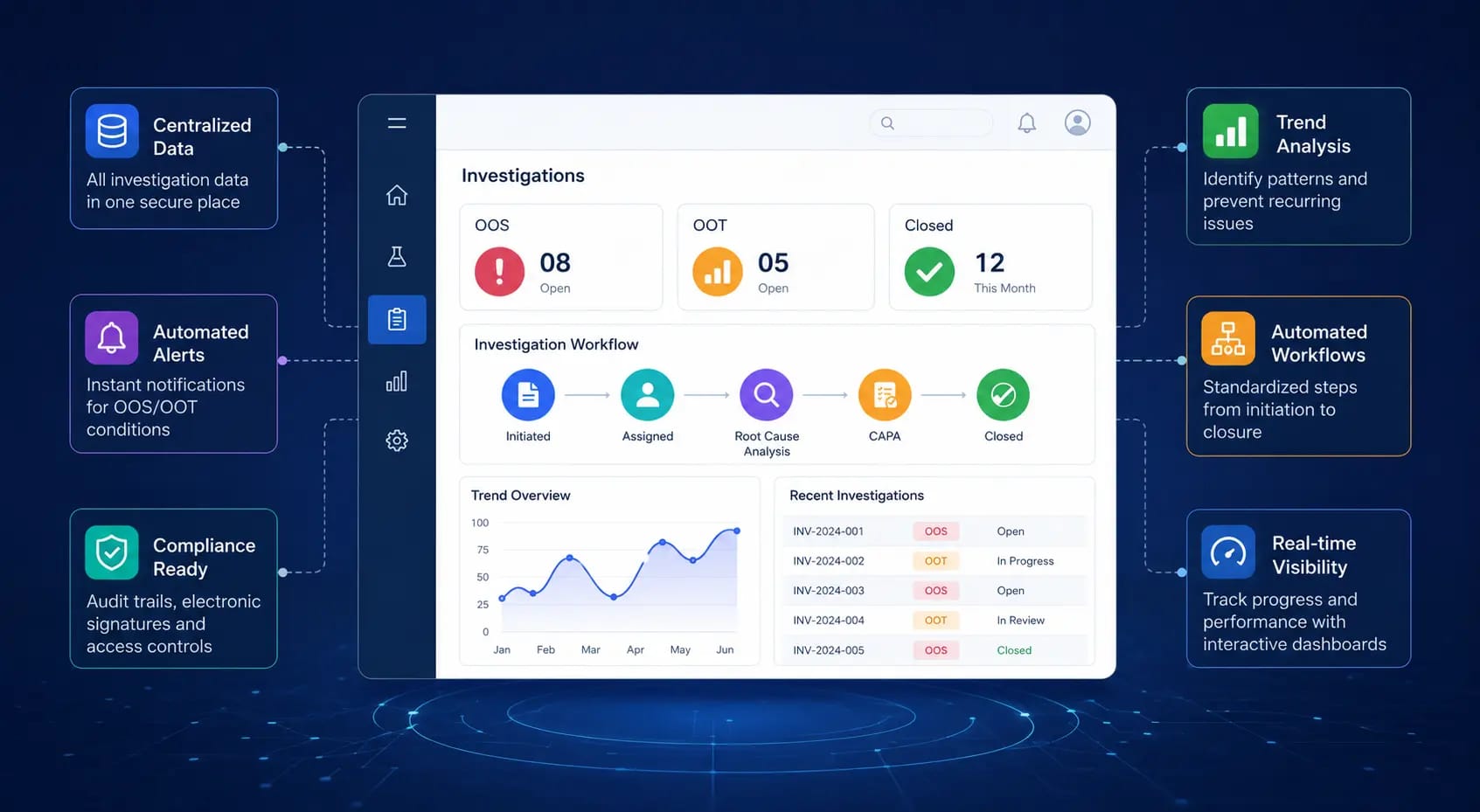
Modern LIMS solutions help pharmaceutical laboratories streamline OOS and OOT investigations through automation, real-time visibility, and improved data integrity. Discover how digital investigation workflows enhance compliance, accelerate root cause analysis, and strengthen overall quality management.

Discover how AI Copilots are transforming Pharmaceutical Quality Management by enabling smarter compliance, faster investigations, predictive insights, and intelligent automation across quality operations. Learn how AI-powered pharmaceutical software helps organizations improve efficiency, reduce manual effort, and build future-ready digital quality systems.

Discover how AI-powered Cleaning Validation Software is helping pharmaceutical manufacturers automate MACO calculations, improve GMP compliance, and eliminate manual validation inefficiencies. Learn how digital validation workflows enhance data integrity, audit readiness, and operational efficiency across pharma manufacturing.

Meet the visionary jury members of AL Ideathon 2024 who evaluated groundbreaking ideas, innovation strategies, and transformative solutions shaping the future of technology and business.


Application Platform as a Service (aPaaS) is a cloud computing model that provides a complete environment for building, deploying, and managing applications without handling underlying infrastructure. It empowers businesses to accelerate development, reduce costs, and scale applications efficiently. By offering tools, frameworks, and automation, aPaaS simplifies the entire application lifecycle from design to deployment helping organizations innovate faster and stay competitive in a digital-first world.

GAMP 5 provides a structured approach for validating computerized systems in the pharmaceutical industry, ensuring they are fit for intended use and aligned with regulatory expectations. It works alongside frameworks like the V-Model, which supports a lifecycle-based validation process from requirements to testing. Regulations such as 21 CFR Part 11 and EU Annex 11 define strict rules for electronic records and signatures, ensuring data integrity, security, and traceability across systems. Together, these standards create a strong compliance ecosystem where organizations can develop, implement, and maintain validated software systems efficiently. By combining risk-based validation, lifecycle management, and regulatory alignment, companies can reduce compliance risks, improve product quality, and accelerate digital transformation in pharma operations

A low-code platform enables organizations to design, develop, and deploy applications with minimal manual coding using visual interfaces and pre-built components. For pharmaceutical companies, it offers a faster and more compliant way to digitize processes, automate workflows, and scale operations while maintaining regulatory standards and data integrity.

Batch Record (BMR) issuance and batch number generation software helps pharmaceutical companies automate and control critical manufacturing processes. It ensures accurate batch tracking, reduces manual errors, and maintains compliance with regulatory standards by providing secure, traceable, and efficient digital workflows.

Pharmaceutical QA/QC batch process automation enables companies to streamline quality assurance and control activities while ensuring compliance with USFDA, MHRA, and cGMP standards. By digitizing workflows and automating critical processes, organizations can enhance data integrity, reduce manual errors, and achieve faster batch release.

A low-code application development platform enables organizations to quickly design, build, and deploy applications using visual tools and minimal coding. In the pharmaceutical industry, it supports faster innovation, improved compliance, and efficient workflow automation while maintaining data integrity and regulatory standards.

A low-code aPaaS (Application Platform as a Service) platform provides a cloud-based environment for rapidly building, deploying, and managing applications with minimal coding. It enables pharmaceutical organizations to streamline workflows, ensure compliance, and scale operations efficiently while accelerating digital transformation.

GMP SOP training planner, management, and tracking software helps pharmaceutical companies efficiently manage employee training programs while ensuring regulatory compliance. By automating training schedules, tracking progress, and maintaining audit-ready records, organizations can improve workforce competency and streamline compliance processes.

CAPA management software helps pharmaceutical organizations efficiently manage corrective and preventive actions to address deviations and improve quality processes. By automating workflows and ensuring traceability, it enhances compliance, reduces risks, and supports continuous improvement.

Change control automation and tracking software helps pharmaceutical organizations manage, document, and track changes efficiently while ensuring regulatory compliance. By automating workflows and maintaining audit trails, it improves traceability, reduces risks, and enhances quality management processes.

QMS automation software helps pharmaceutical and biotech companies streamline quality processes, manage compliance, and improve operational efficiency. By automating workflows such as CAPA, deviations, audits, and document control, it ensures consistent quality and regulatory adherence.

An Exchange Server management tool helps organizations efficiently manage, monitor, and secure their email infrastructure. By automating administrative tasks, tracking performance, and enhancing security, it ensures reliable communication and streamlined IT operations.

A no-code application development platform allows users to build and deploy applications without writing code using visual interfaces and pre-built components. In the pharmaceutical industry, it enables faster innovation, improved compliance, and efficient workflow automation.

Excel automation simplifies repetitive tasks by using tools, macros, and workflows to improve efficiency and accuracy. For pharmaceutical and enterprise environments, it enables faster data processing, reduces manual errors, and enhances productivity through streamlined operations.

An Environmental Monitoring System (EMS) is essential for pharmaceutical and biotech facilities to monitor critical environmental conditions such as temperature, humidity, pressure, and microbial contamination. It ensures product quality, regulatory compliance, and risk reduction by providing real-time data, automated alerts, and complete traceability across manufacturing environments.

A Quality Suite is an integrated platform that helps pharmaceutical and biotech companies manage quality processes such as CAPA, deviations, audits, and document control. By automating workflows and ensuring compliance, it improves efficiency, data integrity, and overall product quality.

An asset management system helps organizations track, manage, and optimize the lifecycle of assets, from procurement to maintenance and disposal. In pharmaceutical and enterprise environments, it ensures operational efficiency, reduces downtime, and supports regulatory compliance.

Process validation software helps pharmaceutical companies ensure that manufacturing processes consistently produce quality products. By automating validation workflows and maintaining detailed documentation, it enhances compliance, improves data integrity, and supports efficient lifecycle management.

Struggling with delays, manual spreadsheets, or lack of traceability in your APQR process? These are clear signs your Annual Product Quality Review may not be compliance ready. Discover the top 5 warning signals and learn how digital APQR systems can improve efficiency, ensure audit readiness, and strengthen regulatory compliance in pharmaceutical operations.

Struggling with inefficiencies in your APQR process? Common mistakes like incomplete data, lack of standardization, and poor traceability can impact compliance and decision-making. Learn how to avoid these pitfalls and improve your Annual Product Quality Review with a structured, digital approach.

Struggling to stay audit-ready in pharma? Manual APQR processes often lead to delays, poor traceability, and compliance risks. Discover how digital APQR systems improve audit readiness with centralized data, real-time visibility, automated documentation, and complete audit trails.


















































FDA Stage 3 Continued Process Verification (CPV) is a critical phase in the process validation lifecycle that ensures manufacturing processes remain in a validated state during commercial production. It focuses on continuous monitoring of critical process parameters (CPPs) and critical quality attributes (CQAs), using statistical tools and real-time data analysis to detect trends, variability, and potential deviations. By adopting a lifecycle approach, the FDA emphasizes that validation is not a one-time activity but an ongoing process driven by data and risk management. CPV integrates process performance data into annual product reviews and management decisions, enabling proactive quality assurance and regulatory compliance. With advanced digital solutions like AmpleLogic, organizations can streamline CPV implementation, enhance data integrity, and ensure continuous compliance with evolving FDA expectations.


A 21 CFR Part 11 compliant document management system is essential for pharmaceutical and life sciences organizations transitioning to digital operations. 21 CFR Part 11 establishes the criteria under which electronic records and electronic signatures are considered trustworthy, reliable, and equivalent to paper-based documentation. By implementing a compliant document management system, organizations can securely create, store, retrieve, and manage regulated documents while maintaining complete audit trails, controlled access, and validated workflows. These systems help ensure data integrity, prevent unauthorized changes, and support regulatory inspections by providing traceable and tamper-proof records. With solutions like AmpleLogic, companies can digitize document control processes, automate approvals using electronic signatures, and achieve seamless compliance with FDA regulations—ultimately improving operational efficiency, reducing compliance risks, and ensuring audit readiness across quality and regulatory functions.

The FDA 510(k) clearance process is a critical regulatory pathway for medical device manufacturers seeking to enter the U.S. market. Also known as premarket notification, it requires companies to demonstrate that their device is “substantially equivalent” to an already legally marketed predicate device in terms of safety and effectiveness. Primarily applicable to Class II and some Class I devices, the 510(k) process involves submitting detailed documentation, including device descriptions, intended use, labeling, risk analysis, and performance testing data. The FDA evaluates this information to determine whether the device meets regulatory standards for clearance and commercial distribution. By adopting digital and automated compliance solutions like AmpleLogic, manufacturers can streamline documentation management, ensure data integrity, and accelerate the 510(k) submission lifecycle—helping reduce delays, improve audit readiness, and bring innovative medical devices to market faster while maintaining regulatory compliance.

Digitalizing laboratory planning and scheduling is essential for modern QC labs facing increasing workloads, complex testing requirements, and strict compliance standards. Traditional approaches—often reliant on spreadsheets, whiteboards, or basic systems—struggle to manage the growing complexity of laboratory operations, where thousands of tests must be coordinated efficiently. By adopting digital solutions, laboratories can significantly improve efficiency, reduce turnaround times, and optimize resource utilization. Proven benefits include faster test execution, reduced inventory requirements, and improved adherence to priority-based testing workflows. Key best practices include implementing digital twin modeling for simulation, prioritizing critical path testing, leveraging campaign-based execution, integrating resource planning, and enabling real-time adaptive scheduling. These strategies provide better visibility, minimize bottlenecks, and enhance decision-making. With platforms like AmpleLogic, organizations can integrate intelligent scheduling, AI-driven insights, and real-time monitoring to transform laboratory operations—ensuring compliance, improving productivity, and building a scalable foundation for continuous improvement in life sciences environments.

Batch release is one of the most critical and highly regulated stages in pharmaceutical manufacturing, requiring thorough review and approval of all production and quality data before product distribution. Traditional manual review processes are often time-consuming, error-prone, and difficult to track—leading to delays and compliance risks. AmpleLogic’s APQR software addresses these challenges by introducing an automated Batch Release Checklist that standardizes and streamlines the entire review process. It integrates data from batch manufacturing records (BMRs), deviations, CAPAs, and quality systems into a structured, audit-ready workflow, ensuring completeness and traceability at every step. With configurable SOP-driven checklists, automated validation flags, digital signatures, and real-time dashboards, the platform enables QA teams to detect issues early, reduce manual effort, and accelerate batch approvals. This results in improved data integrity, enhanced compliance, and faster time-to-market—making batch release more efficient, reliable, and inspection-ready.

FDA warning letters are issued when pharmaceutical companies fail to comply with regulatory standards such as Current Good Manufacturing Practices (CGMP), which are critical for ensuring product quality and patient safety. These letters often highlight recurring compliance gaps that can lead to product recalls, operational disruptions, and reputational damage if not addressed proactively. Common issues identified in FDA warning letters include inadequate raw material testing, poor supplier qualification, lack of proper documentation and quality oversight, insufficient process and cleaning validation, and failures in laboratory controls and data integrity. Additionally, deficiencies in stability testing, batch record management, environmental monitoring, and change control processes are frequently cited by regulators. To address these challenges, organizations are increasingly adopting digital quality management systems, LIMS, and automated workflows that ensure traceability, enforce compliance, and reduce human error. By implementing integrated digital solutions like those offered by AmpleLogic, companies can proactively identify risks, streamline quality processes, and maintain continuous audit readiness—ultimately avoiding costly FDA warning letters and ensuring long-term regulatory compliance.

Food and beverage laboratories play a critical role in ensuring product safety, quality, and regulatory compliance across the production lifecycle. From raw material testing to finished product validation, these labs must manage complex workflows, large data volumes, and stringent food safety standards. LIMS software for food and beverage labs provides a centralized platform to streamline sample tracking, automate data capture, and ensure compliance with global regulations such as ISO 17025, HACCP, and food safety standards. It enables end-to-end traceability—from ingredient sourcing to final product release—while reducing manual errors and improving data integrity. With advanced capabilities like batch and lot traceability, instrument integration, automated workflows, and real-time reporting, modern LIMS solutions empower laboratories to enhance operational efficiency, accelerate testing cycles, and maintain audit readiness. By adopting platforms like AmpleLogic, organizations can transform food quality management processes and ensure consistent, compliant, and high-quality product outcomes.

As pharmaceutical and life sciences organizations face increasing regulatory pressure and operational complexity, the debate between paper-based QMS and digital QMS has become more relevant than ever. Traditional paper-based systems, once the standard, now struggle with inefficiencies such as manual documentation, slow approvals, and difficulty in maintaining audit readiness. Paper-based QMS often leads to risks like document misplacement, version control issues, and limited visibility into quality processes—making compliance management more challenging in highly regulated environments. In contrast, a digital QMS (eQMS) centralizes all quality processes, automates workflows, and ensures real-time access to accurate and traceable data. Digital QMS solutions offer significant advantages, including automated document control, secure audit trails, electronic signatures, and real-time reporting dashboards. These capabilities enhance data integrity, reduce human errors, and improve overall operational efficiency while ensuring compliance with regulations such as FDA 21 CFR Part 11 and GMP standards. With platforms like AmpleLogic, organizations can transition from reactive, manual quality management to a proactive, data-driven approach—enabling faster decision-making, improved compliance, and scalable quality operations. In today’s digital era, a modern QMS is not just an upgrade but a strategic necessity for maintaining competitiveness and regulatory excellence.

Managing documentation in the life sciences industry is complex due to stringent regulatory requirements, high data volumes, and the need for complete traceability. A compliance document management system (DMS) helps organizations digitize, control, and standardize document processes while ensuring adherence to global regulations such as FDA 21 CFR Part 11, EU Annex 11, and GxP guidelines. Traditional paper-based or fragmented systems often lead to inefficiencies, version control issues, and compliance risks. In contrast, a modern electronic document management system provides centralized control over document lifecycles—from creation and review to approval, release, and archival—ensuring accuracy, consistency, and audit readiness. With advanced features such as electronic signatures, audit trails, automated workflows, and AI-powered search, platforms like AmpleLogic enable life sciences organizations to improve data integrity, enhance collaboration, and reduce manual effort. By digitizing document control processes, companies can achieve faster approvals, better compliance visibility, and a scalable foundation for regulatory excellence in highly regulated environments.

Pharmaceutical companies face increasing pressure to reduce IT costs while maintaining compliance, innovation, and operational efficiency. Rising infrastructure expenses, legacy systems, and complex regulatory requirements make IT one of the most significant cost centers in the industry. Strategic IT cost reduction focuses on optimizing—not just cutting—expenses by improving system efficiency, eliminating redundancies, and leveraging modern technologies. For example, consolidating IT infrastructure, rationalizing applications, and outsourcing support services can significantly reduce operational overhead. Studies show that streamlining IT systems and vendor management alone can reduce total IT costs by around 30% within a short period. Key strategies include adopting cloud-based platforms, automating manual processes, integrating quality and compliance systems, and reducing dependency on multiple disconnected tools. Additionally, outsourcing specialized functions and improving resource utilization can drive substantial efficiency gains, with some organizations achieving dramatic cost savings through these combined approaches. With platforms like AmpleLogic, pharma companies can unify systems, automate workflows, and enhance data visibility—enabling smarter decision-making and scalable digital transformation. By implementing the right mix of automation, consolidation, and process optimization, organizations can significantly lower IT costs while improving compliance, agility, and overall business performance

Conducting an effective Annual Product Quality Review (APQR) is critical for ensuring consistent product quality and regulatory compliance in the pharmaceutical industry. However, organizations often face multiple challenges that make the process complex, time-consuming, and prone to errors. One of the primary challenges is fragmented data spread across systems like LIMS, QMS, ERP, and spreadsheets, making data aggregation difficult and error prone. Additionally, manual data handling leads to inconsistencies, duplication, and data integrity issues, which can compromise the accuracy of APQR reports. Other common challenges include regulatory compliance pressure, limited analytical capabilities for trend analysis, inefficient cross-functional collaboration, and lack of traceability during audits. These issues often result in delays, increased manual effort, and reduced effectiveness of the review process. To overcome these challenges, pharmaceutical companies are adopting digital solutions that integrate data sources, automate workflows, and provide real-time analytics. Implementing standardized SOPs, improving data governance, enabling collaboration across departments, and leveraging advanced APQR software can significantly enhance efficiency, ensure compliance, and transform APQR into a proactive, data-driven quality process.

Software as a Medical Device (SaMD) refers to software intended to perform medical functions—such as diagnosis, treatment, or monitoring—without being part of a physical medical device. According to global regulatory bodies like the International Medical Device Regulators Forum (IMDRF) and the FDA, SaMD operates on general-purpose platforms such as computers, mobile devices, or cloud environments while delivering clinically relevant outcomes. SaMD solutions are widely used across healthcare, from diagnostic imaging analysis and clinical decision support systems to mobile health applications that monitor patient conditions. These systems must comply with strict regulatory requirements, including risk classification, validation, cybersecurity, and data integrity standards to ensure patient safety and effectiveness. With the rapid growth of digital health and AI-driven technologies, SaMD is transforming how healthcare is delivered by enabling faster diagnosis, personalized treatment, and real-time monitoring. However, it also introduces challenges related to compliance, quality assurance, and lifecycle management. Platforms like AmpleLogic help organizations streamline validation, ensure regulatory compliance, and manage the complete lifecycle of medical device software—enabling faster innovation while maintaining safety and compliance standards.

A Quality Management System (QMS) for medical devices is a structured framework of processes, procedures, and responsibilities designed to ensure product quality, safety, and regulatory compliance throughout the entire lifecycle—from design and development to manufacturing and post-market surveillance. Medical device manufacturers must comply with stringent global regulations such as ISO 13485, EU MDR, and the FDA’s Quality Management System Regulation (QMSR), which aligns with international standards to ensure consistent product quality and patient safety. A robust QMS integrates critical elements such as design controls, risk management, supplier quality management, CAPA (Corrective and Preventive Actions), training management, and audit processes. These components help organizations maintain traceability, minimize risks, and ensure continuous improvement across operations. By adopting digital QMS solutions like AmpleLogic, medical device companies can streamline quality processes, automate compliance workflows, and improve data integrity. This enables faster regulatory approvals, enhanced audit readiness, and consistent delivery of safe, high-quality medical devices in a highly regulated industry.

Artificial Intelligence is rapidly transforming Quality Management Systems (QMS) in the pharmaceutical and life sciences industry by addressing inefficiencies in traditional, manual processes. Conventional QMS often struggle with fragmented data, delayed investigations, and limited visibility across quality events—impacting compliance and operational efficiency. By integrating AI into QMS, organizations can automate critical quality processes such as CAPA, deviation management, change control, and audit management. AI-powered systems leverage machine learning and predictive analytics to identify trends, detect anomalies, and recommend corrective actions—enabling faster root cause analysis and proactive risk mitigation. AI-driven QMS also enhances decision-making by providing real-time insights, improving data accuracy, and reducing manual intervention. Capabilities such as predictive OOS/OOT detection, automated complaint handling, and intelligent risk assessment help organizations improve product quality, ensure regulatory compliance, and reduce operational costs. With platforms like AmpleLogic, pharmaceutical companies can adopt a fully integrated, AI-powered quality ecosystem—streamlining workflows, accelerating approvals, and achieving up to significant efficiency gains while maintaining strict compliance with global regulations.

Medical device document control is a critical component of quality and regulatory compliance, ensuring that all documents related to design, manufacturing, and quality management are properly created, reviewed, approved, and maintained throughout their lifecycle. It plays a vital role in maintaining product safety, traceability, and audit readiness in highly regulated environments. Regulatory frameworks such as FDA 21 CFR Part 820, ISO 13485, and EU MDR require organizations to establish formal procedures for document approval, version control, distribution, and change management. These regulations ensure that only current, approved documents are in use and that all changes are tracked and validated to maintain data integrity. A robust document control system enables organizations to manage critical records such as Device Master Records (DMR), Device History Records (DHR), and quality procedures with full traceability. By adopting digital solutions like AmpleLogic, companies can automate document workflows, enforce compliance, maintain secure audit trails, and improve operational efficiency—ensuring readiness for regulatory inspections and long-term quality excellence.

Quality Key Performance Indicators (KPIs) are essential metrics used to evaluate the effectiveness of quality management systems (QMS) in pharmaceutical manufacturing. These KPIs provide measurable insights into product quality, process efficiency, and regulatory compliance—helping organizations make data-driven decisions and ensure continuous improvement. In pharma manufacturing, commonly tracked KPIs include batch rejection rate, deviation rate, CAPA effectiveness, out-of-specification (OOS) incidents, and cycle time for investigations. These indicators help identify process inefficiencies, detect quality issues early, and ensure timely corrective actions. Monitoring such KPIs is critical for maintaining compliance with GMP standards and ensuring consistent product quality. Additionally, metrics such as Right First Time (RFT), defect rate, and process cycle time provide insights into manufacturing performance and operational efficiency. By analyzing these KPIs, organizations can reduce rework, minimize costs, and improve overall production outcomes. With digital platforms like AmpleLogic, pharma companies can automate KPI tracking, generate real-time dashboards, and gain predictive insights—transforming quality management into a proactive, data-driven function that enhances compliance, efficiency, and product reliability.

Stability studies are a critical component of pharmaceutical development, ensuring that drug products maintain their identity, strength, quality, and purity throughout their shelf life under defined environmental conditions. These studies help establish expiration dates, storage requirements, and regulatory compliance in line with ICH guidelines. Regression analysis plays a vital role in interpreting stability data by modeling degradation patterns and predicting product shelf life. By analyzing trends influenced by factors such as temperature, humidity, and light exposure, regression techniques enable accurate estimation of expiry dates and support data-driven decision-making in pharmaceutical quality management. Advanced statistical methods like ANOVA and ANCOVA further enhance stability analysis by identifying significant variations across conditions and adjusting for external variables. These approaches improve the precision and reliability of stability predictions, ensuring consistent product performance and regulatory compliance. By leveraging digital solutions like AmpleLogic’s stability study management software, organizations can automate workflows, monitor real-time trends, and apply intelligent statistical models—transforming stability testing into a more efficient, accurate, and compliant process that ensures long-term pharmaceutical product reliability.

Deviation management is a critical component of pharmaceutical quality systems, ensuring that any non-conformance is properly identified, investigated, and resolved in compliance with regulatory standards. However, traditional approaches are often slow, manual, and resource-intensive, leading to delays in investigations and increased compliance risks. Artificial Intelligence is transforming deviation management by automating key steps such as data collection, root cause analysis, and CAPA recommendations. AI-powered systems analyze historical deviations, identify patterns across processes and equipment, and provide data-driven insights that help quality teams make faster and more accurate decisions. With AI-enabled solutions like AmpleLogic’s QMS, organizations can significantly reduce investigation timelines, improve CAPA effectiveness, and minimize deviation recurrence. These systems enhance traceability, ensure audit readiness, and enable proactive quality management—shifting from reactive issue handling to predictive, intelligence-driven compliance.

Stability software plays a crucial role in ensuring compliance and maintaining product quality throughout the pharmaceutical development lifecycle. Stability studies are essential for determining how drug products maintain their identity, strength, quality, and purity under various environmental conditions, making them a cornerstone of regulatory approval and lifecycle management. Modern stability management software enables organizations to design, execute, and monitor ICH-compliant stability studies efficiently. These systems automate critical processes such as protocol management, sample pull scheduling, environmental monitoring, and statistical trend analysis—ensuring accurate shelf-life prediction and robust regulatory submissions. By digitizing stability workflows, pharmaceutical companies can eliminate manual tracking, improve data integrity, and maintain complete audit trails aligned with FDA 21 CFR Part 11 and GxP requirements. Advanced platforms also integrate with LIMS and QMS systems, providing real-time visibility into study progress and enabling proactive decision-making. With solutions like AmpleLogic, organizations can streamline stability study management, enhance compliance, and ensure consistent product quality—ultimately accelerating development timelines and improving overall operational efficiency in a highly regulated environment.























Annual Product Quality Review (APQR) in pharma is a GMP requirement that evaluates product quality, manufacturing consistency, deviations, and compliance over time to ensure continuous improvement and regulatory adherence.

Top challenges in Product Quality Review (APQR) in pharma include data integration issues, inconsistent documentation, deviation management, regulatory compliance gaps, and limited trend analysis—impacting quality, efficiency, and continuous improvement.

Manual vs automated APQR in pharma explore key differences in efficiency, data accuracy, compliance, and scalability to help pharma leaders choose the right approach for quality review and continuous improvement.

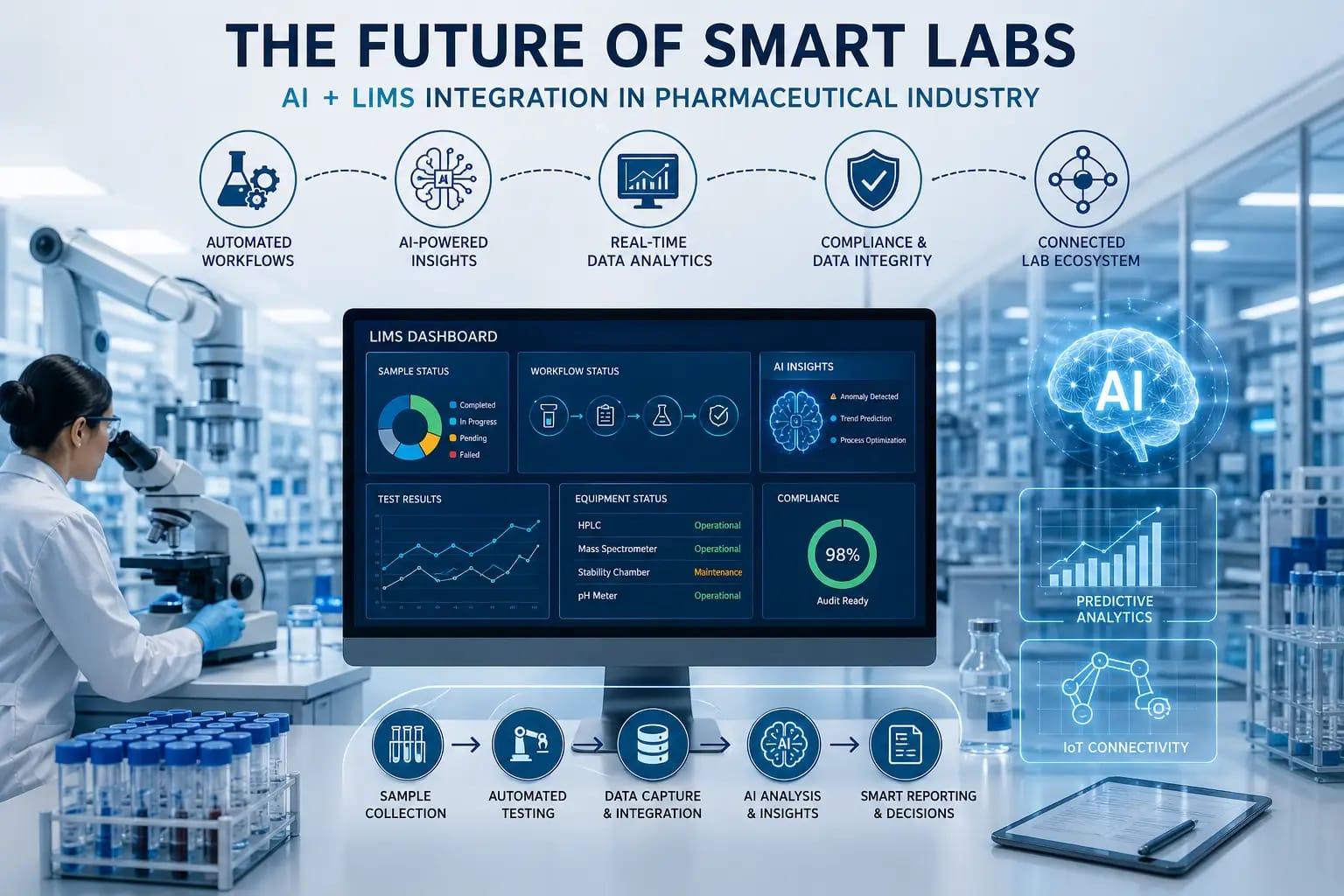
AmpleLogic’s inclusion in the Gartner Market Guide for Laboratory Information Management Systems (LIMS) underscores its growing influence in the life sciences technology space. With its low-code, AI-powered platform, AmpleLogic enables laboratories to streamline workflows, enhance data integrity, and accelerate digital transformation initiatives. This recognition reflects the company’s commitment to delivering innovative, compliant, and scalable solutions for modern laboratories.

Automating batch release within the APQR process transforms how pharmaceutical companies manage quality and compliance. Traditionally, batch release involves extensive manual review of manufacturing and quality records to ensure a product is fit for distribution. Annual Product Quality Review (APQR) consolidates data from all batches over a year to identify trends and ensure consistent product quality. By integrating automation into APQR, organizations can digitize batch review workflows, implement checklist-driven validations, and enable real-time tracking of deviations, CAPAs, and quality metrics. This not only improves data integrity and audit readiness but also significantly reduces release cycle times. With platforms like AmpleLogic, pharma companies can shift from reactive quality checks to proactive, data-driven batch release decisions, ensuring faster approvals without compromising compliance.














A comprehensive guide to investigating Out-of-Specification (OOS) results in analytical testing. Learn structured workflows, root cause analysis, and regulatory-compliant approaches using AmpleLogic’s eQMS to ensure data integrity, quality assurance, and effective CAPA management in regulated industries.

Learn how AmpleLogic’s eQMS simplifies the lifecycle of a Quality Management System in the pharmaceutical industry. From deployment to continuous improvement, achieve seamless compliance, improve product quality, and digitize critical quality processes with an advanced, scalable platform.

Human errors in life sciences can lead to compliance risks and costly deviations. Explore six essential digital tools that streamline workflows, automate data capture, and ensure accuracy across pharmaceutical and biotech operations.

Human error remains one of the leading causes of cybersecurity breaches, accounting for up to 95% of incidents across industries. In life sciences, even minor mistakes can compromise sensitive data, disrupt compliance, and impact patient safety—making robust digital systems essential.

Explore how Cipla streamlined user access management in its pharmaceutical operations using AmpleLogic’s advanced UAM solution. This case study highlights improved compliance, automated user provisioning, and secure, audit-ready access control aligned with GxP requirements.

Understand the regulatory requirements for MACO calculation and cleaning validation in pharmaceutical manufacturing. This guide explains how global agencies like FDA, EMA, CDSCO, and WHO expect scientifically justified, risk-based residue limits using PDE and dose-based approaches to ensure compliance, patient safety, and audit readiness.

Human error remains one of the leading causes of compliance issues in pharmaceutical manufacturing. By adopting digital solutions such as eBMR, QMS, and LIMS, organizations can eliminate manual inefficiencies, improve data accuracy, and ensure regulatory compliance through automation, real-time monitoring, and standardized workflows.

Explore how Teva leveraged AmpleLogic’s eQMS to digitize quality processes, improve compliance, and reduce manual workloads. This case study highlights how a unified digital platform enabled faster operations, better audit readiness, and scalable quality management in the pharmaceutical industry.

Outdated application software can expose organizations to significant security risks, compliance failures, and operational inefficiencies. This article explores how legacy systems impact regulated industries like pharmaceuticals and why upgrading to modern, secure digital solutions is essential. Learn how AmpleLogic helps businesses stay compliant, secure, and future-ready.

AmpleLogic’s LMS for the cosmetic industry streamlines training, ensures regulatory compliance, and enhances workforce competency. With AI-driven automation, SOP management, and GMP-ready features, it empowers cosmetic companies to maintain quality, safety, and audit readiness in a highly regulated environment.

The FDA software validation process is essential for ensuring that computerized systems in pharma consistently perform as intended while meeting regulatory requirements. This blog explains validation lifecycle stages, documentation, risk-based approaches, and how digital tools help achieve compliance, data integrity, and audit readiness.

AmpleLogic’s QC Planning & Scheduling Software empowers pharmaceutical laboratories with intelligent planning, automated scheduling, and campaign-based execution. It enhances resource utilization, reduces turnaround time, and ensures compliance through real-time visibility and optimized laboratory workflows.

A practical eLogbook checklist helps QA, QC, and production teams standardize documentation, reduce human errors, and maintain GMP compliance. By digitizing logbooks, pharma companies can improve data integrity, ensure audit readiness, and streamline daily operational workflows.

Learn how OCuSOFT leveraged AmpleLogic’s eQMS to digitize end-to-end quality processes, reducing paper-based records by 85% while improving visibility, compliance, and audit readiness. The solution streamlined document control, CAPA, and audit workflows in a regulated environment.

Responding to FDA 483 observations requires a timely, structured, and evidence-based approach. Organizations must address each observation with root cause analysis, corrective and preventive actions (CAPA), and clear timelines to avoid warning letters and strengthen compliance.

Excluding QA from facility changes can lead to compliance gaps, validation failures, and increased regulatory risk. QA plays a critical role in assessing impact, ensuring proper documentation, and maintaining GMP standards through structured change control and risk management processes.

Pharmaceutical batch issue procedures are critical for ensuring GMP compliance, traceability, and product quality. From issuing Batch Manufacturing Records (BMR) to verifying raw materials and documenting every step, these processes provide a complete audit trail for each batch and support regulatory inspections.

Incomplete batch records are a major red flag during FDA audits, often leading to 483 observations and warning letters. Missing signatures, unclear entries, and data gaps raise concerns about product quality, traceability, and data integrity, making robust documentation practices essential for compliance.
















Explore how ADC Therapeutics digitized standard management with AmpleLogic to improve compliance, reduce manual effort, and enhance data visibility and efficiency.

Learn the common MHRA data integrity failures in pharma and how to prevent them. Improve compliance, ensure ALCOA+ principles, and stay audit-ready with best practices.

Discover how sterile LIMS differs from OSD LIMS in pharma. Compare workflows, compliance needs, and data management to choose the right solution.

Understand how human error impacts data integrity in pharma. Learn FDA compliance requirements, ALCOA+ principles, and proven strategies to prevent audit failures.

Learn how to investigate OOS (Out of Specification) results in analytical testing. Understand root cause analysis, FDA guidelines, and CAPA strategies for pharma compliance.


















Explore how AI and no-code platforms are transforming pharma manufacturing with smarter quality control, predictive maintenance, and inspection-ready documentation.

Discover the hidden operational debt pharma companies face by delaying low-code and AI adoption—and how AmpleLogic helps reduce it with GxP-compliant workflows.

Automate Annual Product Quality Reviews with AmpleLogic. Reduce prep time by 70–80%, ensure GMP compliance, and generate regulatory-ready PQR reports faster.

Learn why pharma regulatory teams need a RIMS solution to manage submissions, renewals, CMC updates, and global filings accurately with AmpleLogic RIMS.

Discover why pharma plants need eLogbooks to ensure compliance, eliminate manual errors, and improve data integrity with AI-powered solutions.

AmpleLogic AI reduces pharma deviation handling time, automates root cause analysis, improves compliance, and speeds up investigations.

AmpleLogic guide to cleaning validation in pharma ensures GMP compliance, prevents contamination, and improves manufacturing efficiency.


Eisai Pharmaceuticals Successfully Went Live with AmpleLogic Employee Training Management Software

Hetero Labs Limited Goes Live with AmpleLogic Calibration and Preventive Maintenance Software













As a pharmaceutical manufacturer, you are likely familiar with the stringent regulatory requirements governing the industry. One critical aspect of compliance is to maintain accurate and up-to-date records and documentation. But have you considered the role of logbooks in your record-keeping system? If not, you may want to start.

Incorporating digital technologies in the life sciences industry has become a business necessity in recent times. With daily regulatory updates, increasing documentation, and the need for faster audits, pharmaceutical companies are steadily moving away from manual processes. One such shift is toward Document Management Systems (DMS), a tool that’s becoming vital for ensuring compliance, efficiency, and data integrity. While digital innovations have touched every sector, their role in pharma operations and quality assurance is especially transformative. Among the many tools transforming the industry, DMS stands out as an important solution, automating document workflows, reducing dependency on paper, and offering centralized control over crucial documentation.

The growing favor of e-learning through Electronic Learning Management Systems software has gained significant traction across various industries, especially in the pharmaceutical sector. As one of the most vital sectors in Europe and other economies, the pharmaceutical industry has seen rapid growth in recent years. After the pandemic, healthcare and vaccination became top priorities for many countries. However, the urgency extended to other essential medicines in the market as well. In this fast-paced environment, pharmaceutical LMS has become a critical feature for training new personnel and offering certifications to ensure that workers are prepared to handle the increasing demands of the sector. As the pharmaceutical industry continues to expand, the need for a skilled workforce is rising steadily. According to the most recent MarketsandMarkets report:

The term Pharma 4.0 is no longer a concept discussed in conferences; it’s being put to work on the ground in India. Across formulation labs and manufacturing units, pharma companies are steadily upgrading outdated systems, bringing in automation, data visibility, and smarter infrastructure. This shift solves local industry problems: production delays, data integrity lapses, manual dependencies, and growing regulatory scrutiny. As the Indian pharma sector embraces digitalization and advanced technologies, several necessary transformations are underway:






















In today’s highly regulated life sciences environment, organizations must balance agility with strict compliance requirements. Low-code QMS and MES solutions are transforming how pharmaceutical and biotech companies manage quality, manufacturing, and regulatory workflows by enabling rapid application development without extensive coding. AmpleLogic’s low-code platform provides a unified ecosystem that integrates quality management, manufacturing execution, laboratory systems, and regulatory processes into a single data-driven framework. This eliminates silos, enhances real-time visibility, and enables faster decision-making across operations. With built-in compliance frameworks such as FDA 21 CFR Part 11, GxP, and Annex 11, organizations can ensure audit readiness while accelerating deployment timelines from months to weeks. The platform’s drag-and-drop configuration, automated workflows, and AI-powered insights help reduce manual effort, improve data integrity, and drive continuous improvement. By adopting low-code QMS and MES solutions, companies can achieve greater operational agility, reduce costs, and maintain consistent regulatory compliance—making it a critical strategy for modern digital transformation in the life sciences industry.

The pharmaceutical industry is on the cusp of a digital revolution. Industry 4.0 technologies are transforming how drugs are manufactured, tested, and distributed globally.

A Laboratory Information Management System (LIMS) is a vital tool for modern pharmaceutical laboratories, streamlining workflows, enhancing compliance, and driving efficiency.

Regulatory compliance in pharmaceutical cleaning validation is non-negotiable. Understanding and implementing the right protocols ensures product safety and manufacturing integrity.

Implementing cleaning validation best practices in pharmaceutical manufacturing is essential for maintaining product quality, preventing cross-contamination, and ensuring GMP compliance.

Selecting the right LIMS software is a critical decision for pharmaceutical laboratories. The right system can transform lab operations, enhance compliance, and accelerate time-to-results.

The integration of Artificial Intelligence and Machine Learning into Manufacturing Execution Systems is transforming pharmaceutical production with predictive analytics and real-time optimization.

Long-range planning is a strategic imperative for pharmaceutical companies navigating complex regulatory landscapes, evolving market dynamics, and technological transformation.

The convergence of aPaaS (Application Platform as a Service) with COTS (Commercial Off-The-Shelf) solutions and low-code development is reshaping how pharmaceutical companies build and deploy applications.

Competition is exceedingly high within the pharmaceutical and biotechnology industry whereby quality standards are thus reprehended. This requires constant supervision and investigation of processes involved.

Continuous monitoring of environmental conditions is paramount in the pharmaceutical industry and various other sectors, ensuring adherence to regulatory standards and product quality.

The pharmaceutical industry is increasingly moving towards paperless Batch Manufacturing Records (BMRs) driven by regulatory expectations and the need for enhanced data integrity and compliance.

Cleaning validation in pharmaceutical industry is an essential process outlined by regulatory bodies to maintain product quality and prevent cross contamination.

In today's fast-paced and highly regulated industries, Product Review software has become an indispensable tool. Evolving from basic tracking systems to sophisticated, integrated platforms.

In the pursuit of operational excellence and quality assurance, deviations from established norms are not just expected but anticipated. What truly differentiates successful organizations is how they handle these deviations.
%20Software.webp&w=3840&q=80)
In today's competitive business landscape, maintaining high product quality and continuously improving offerings is essential for success. APR software has emerged as a powerful tool.
In the dynamic and highly regulated world of pharmaceutical manufacturing, integration of advanced technology is essential to excel in key business processes and ensure standard product quality.
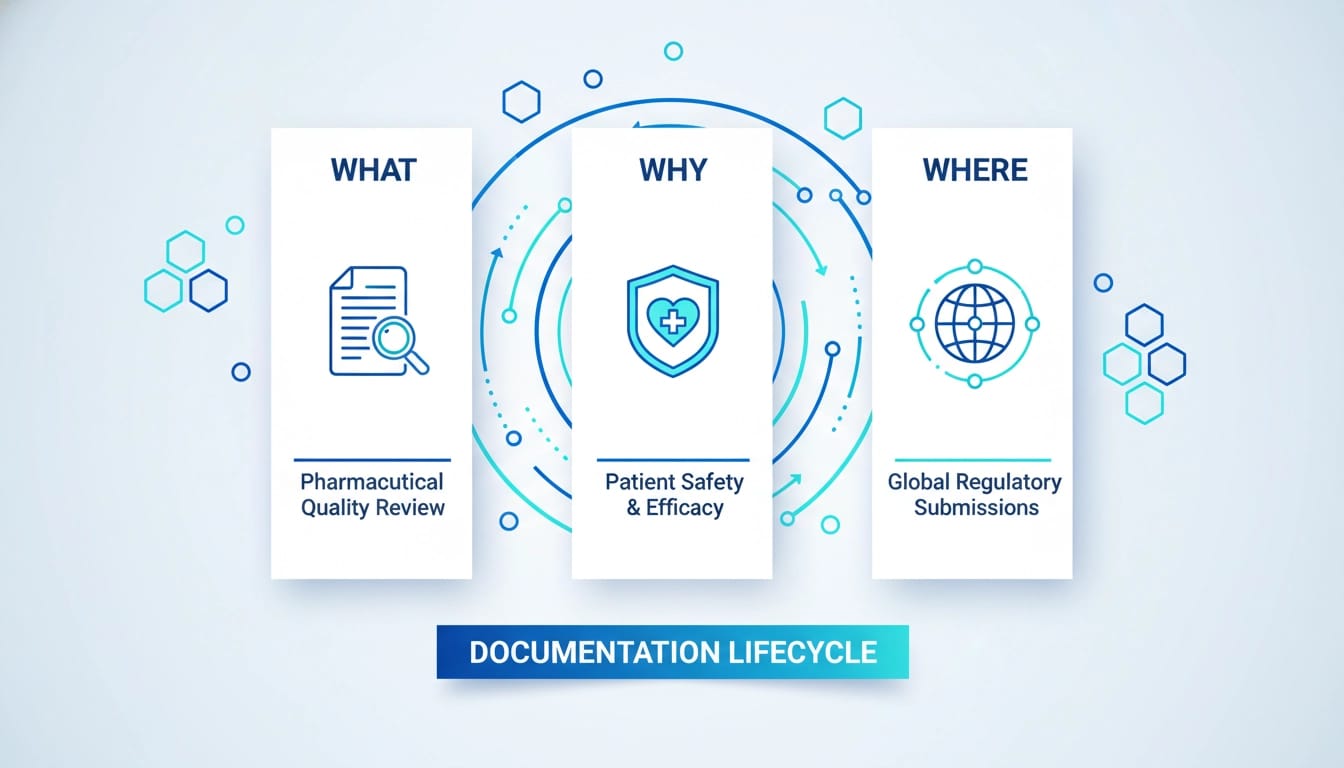
Embarking on an exploration of the essential aspects surrounding Annual Product Quality Review (APQR), this investigation delves into the fundamental questions of What, Why, and Where.

In the pharmaceutical industry, validation of Batch Manufacturing Records (BMR) is critical to ensure product quality, safety, and regulatory compliance. Traditionally, this validation has been a manual process.

In the rapidly evolving landscape of pharmaceuticals, the efficient management of assets is paramount. From research laboratories to manufacturing facilities and distribution networks.

In the pharmaceutical industry, the integrity of data is paramount. The ALCOA principles—Attributable, Legible, Contemporaneous, Original, and Accurate—were established to ensure data integrity in manufacturing.

Learning Management Systems (LMS) are increasingly vital in the pharmaceutical industry, providing a structured and efficient way to manage training, compliance, and professional development.
-in-Equipment.webp&w=3840&q=80)
Efficiency and precision are quintessential aspects in the life sciences and pharmaceutical industries. They help companies attain business process excellence departing from traditional manual processes.

The Annual Product Quality Review (APQR) is a critical process for pharmaceutical companies to evaluate the quality standards of their products, ensure regulatory compliance, and drive continuous improvement.

Change control is a critical process in regulated industries, such as pharmaceuticals, medical devices, and food and beverages. It involves managing changes to products, processes, and systems.

Equipment maintenance is a crucial aspect of ensuring the efficiency and productivity of pharmaceutical production and manufacturing departments. Equipment maintenance software has revolutionized the field.
In the intricate world of pharmaceutical manufacturing, where precision, compliance, and efficiency are key assets, businesses seek innovative solutions to navigate the complexities.

In the intricate and ever-evolving landscape of modern industry, where the quest for operational excellence is both a strategic imperative and a cornerstone of sustained success.

In the pharmaceutical industry, maintaining data integrity is crucial to ensure product quality, patient safety, and regulatory compliance. Data integrity refers to the accuracy and consistency of data.
-Software-in-Deviation-Handling.webp&w=3840&q=80)
Efficiency, accuracy, and compliance are essential components in the pharmaceutical manufacturing industry. Any deviation from standard operating procedures can compromise product quality.

Root Cause Analysis (RCA) is a method used to address a problem or non-conformance in the quality process to get to the "root cause" of the non-conformity. RCA helps to correct or eliminate the cause and prevent the problem from recurring through preventive action.

A pharmaceutical Training Management System (TMS) streamlines GMP compliance training, competency tracking, and regulatory audit readiness. Discover how a modern LMS reduces training costs by up to 50%, accelerates employee qualification, and ensures 21 CFR Part 11 compliance across global operations.

The Electronic Document Management System (eDMS) has fundamentally changed how pharmaceutical and life sciences companies handle documentation. Yet despite FDA 21 CFR Part 11 validation and clear compliance benefits, many organizations still hesitate to fully embrace this digital transformation.

A long list of data integrity warnings received by pharma companies has made them train their focus on building and fortifying their tracking mechanisms. One of the solutions is digitization — but the current challenge is that digital transformation is going slower than expected.
Stay updated with the latest trends in pharma manufacturing, compliance, and digital transformation.
Get the latest product updates, compliance news, and industry insights delivered to your inbox.