Track Every Submission and Registration Globally
Purpose-built regulatory information management system for pharmaceuticals — automate submissions, track ANDA & DMF filings, and manage drug product lifecycles across 120+ countries with AI-powered regulatory compliance software.
120+
Countries Supported
50%
Faster Submissions
100%
Audit-Ready
24/7
Compliance Monitoring
RIMS Challenges: Why Manual Regulatory Management Falls Short
Critical challenges that hinder compliance, submissions, and market access
Constantly Evolving Global Regulations
Keeping pace with FDA, EMA, MHRA, CDSCO, WHO, Health Canada, TGA, and 100+ regional authorities demands continuous vigilance and rapid adaptation.
Fragmented Regulatory Data
Critical regulatory information scattered across emails, spreadsheets, shared drives, and legacy systems creates blind spots and compliance gaps.
Complex Submission Formats
Managing eCTD, ACTD, NeeS, and paper-based submissions across different regions requires specialized expertise and error-prone manual coordination.
Poor Visibility & Tracking
No unified dashboard for real-time status of global submissions, approvals, queries, and renewal timelines across markets.
Manual & Error-Prone Processes
Heavy reliance on email communication, manual data entry, and disconnected spreadsheets increases risk of missed deadlines and regulatory penalties.
Integration Challenges
Disconnected QMS, DMS, ERP, and Safety systems cause data duplication, inconsistencies, and slow cross-functional regulatory workflows.
Scalability Issues
Growing product portfolios across geographies exponentially increase regulatory complexity, overwhelming manual tracking approaches.
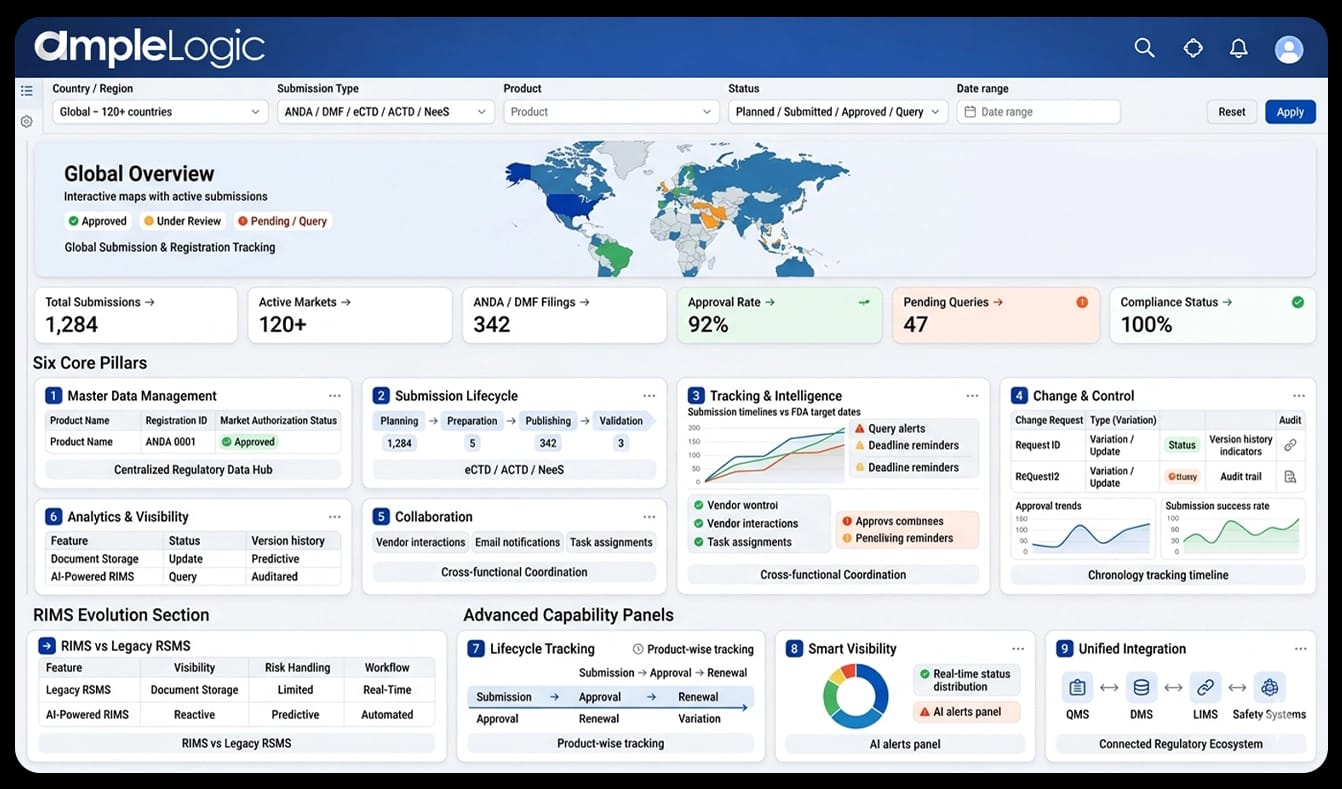
AmpleLogic RIMS Software Overview
Six pillars enabling end-to-end regulatory lifecycle management
Master Data Management
Centralized product registration, global market authorization, and lifecycle tracking across all regulatory authorities.
Submission Lifecycle
Automated workflows for planning, preparation, publishing, and validation of eCTD, ACTD, and NeeS submissions with ANDA and DMF tracking.
Tracking & Intelligence
Real-time submission status, approval and query management, FDA target dates, and proactive deadline monitoring across all markets.
Change & Control
Change control, variation management, and continuous improvement process (CIP) with full audit trails and version history.
Collaboration
Vendor coordination, customer notification, and automated email alerts for seamless cross-functional regulatory workflows.
Analytics & Visibility
Graphical dashboards, chronology tracking, lifecycle analytics, and compliance-ready intelligence for informed regulatory decisions.
RIMS: The Next Evolution of RSMS
While legacy RSMS platforms were passive document stores, RIMS is an intelligent, AI-powered regulatory management ecosystem that actively drives compliance, predicts risks, and accelerates time-to-market.
Data Management
Complete product registration and global regulatory data in a centralized system with master data governance and lifecycle tracking.
Lifecycle Tracking
Monitoring approvals, variations, and renewals to ensure continuous regulatory compliance across all markets.
Smart Visibility
Real-time global visibility and proactive monitoring through intelligent alerts and compliance dashboards.
Unified Integration
Seamless integration with QMS, DMS, LIMS, and Safety systems to build a unified regulatory management ecosystem.
RIMS Workflow Modules: Submission Lifecycle Management
Eight interconnected modules guiding regulatory processes from strategy to ongoing compliance
Regulatory Data Foundation
Global market setup and master data repository for DMF, CEP, LOA, and product lifecycle tracking across all authorities.
Submission Planning
Strategy development, timeline management, milestone tracking, and resource allocation for global regulatory submissions.
Submission Preparation
Dossier compilation, version control, and document assembly for eCTD / ACTD / NeeS readiness with automated validation checks.
Submission Execution
Validation, publishing to health authorities, gateway submission, ANDA tracking, and real-time authority status monitoring.
Review Management
Query tracking, deficiency management, authority communication logs, and response timeline monitoring across all markets.
Drug Product Lifecycle Management
Variations, renewals, post-approval changes, and label updates with complete lifecycle tracking and automated alerts.
Compliance Monitoring
Automated alerts, commitment tracking, inspection readiness, and proactive regulatory change intelligence.
Analytics & Reporting
Real-time dashboards, graphical reports, global submission insights, and GDUFA/authority-specific analytics.

End-to-End Orchestration
Each workflow module integrates seamlessly with QMS, DMS, LIMS, and Safety systems, eliminating data silos and manual handoffs across the entire regulatory lifecycle.
Key Features of AmpleLogic RIMS Software
Twelve critical capabilities enabling regulatory excellence
Product Registration Tracking
Track product registrations across 120+ countries with automated status updates, deadline alerts, and complete drug product lifecycle management.
Submission Preparation & Publishing
Approval & Rejection Monitoring
ANDA Tracker & FDA Target Dates
DMF Tracker & Change Control
Commitment & Variation Management
Vendor & Stakeholder Management
Graphical Reports & Dashboards
Automated Pharma Regulatory Workflows
Electronic Signatures & Audit Trails
Centralized Document Repository
Submission Validation Engine
AI-Powered RIMS: Intelligent Regulatory Analytics
Six AI-powered capabilities accelerating regulatory workflows and reducing risk
AI Compliance Tracking
Continuously monitors and adapts to evolving local and global regulatory standards, automatically updating processes to ensure compliance without manual intervention.
Predictive Analytics
Analyzes historical and real-time data to identify potential risks and compliance gaps before they escalate, helping proactively mitigate issues and avoid penalties.
Automated Reporting
AI automates the generation of regulatory reports and documentation, ensuring they are accurate, timely, and audit-ready with minimal human involvement.
Risk Mitigation Engine
AI-powered algorithms assess data patterns to flag potential compliance risks, offering predictive insights and recommending corrective actions.
Regulatory Change Intelligence
Tracks and processes regulatory changes in real time, automatically implementing them into your systems to ensure ongoing compliance with latest standards.
Workflow Optimization
AI optimizes workflows, minimizing operational costs, speeding up regulatory processes, and ensuring consistent compliance across all business levels.
Global Regulatory Trackers: FDA, EMA & RoW Monitoring
Three regional tracking modules for comprehensive market authorization
USA FDA Regulatory Tracker
ANDA tracker and DMF tracker with FDA lifecycle milestones including IND, NDA, ANDA, BLA target action dates, GDUFA fees, and 505(b)(2) submissions.
EU & UK Regulatory Tracker
Manage EMA and MHRA procedures, centralized/decentralized/mutual recognition variations, PSUR schedules, and post-Brexit UK submissions with automated workflows.
Global RoW Regulatory Tracker
Streamline APAC, LATAM, MENA, and Africa submissions with country-specific lifecycle tracking for Health Canada, TGA, CDSCO, WHO PQ, and 100+ authorities.
RIMS Compliance: Global Regulatory Standards
Full support for global regulatory frameworks and compliance standards
Major Authority Support
Full coverage for US FDA, EMA, MHRA, WHO, Health Canada, TGA, CDSCO, ANVISA, PMDA, and 100+ global health authorities.
Multi-Region Format Management
Native support for eCTD, ACTD, NeeS, and paper-based formats with automated renewal monitoring and format-specific validation.
21 CFR Part 11 & EU Annex 11
Fully compliant controls including electronic signatures, audit trails, data integrity safeguards, and secure version control.
Proactive Monitoring & Alerts
Automated alerts for deadlines, commitment due dates, renewal windows, and regulatory changes with real-time risk reduction.
IDMP Readiness
Supports ISO IDMP standards for substance, product, organization, and referential data to prepare for EMA's identification requirements.
ICH eCTD 4.0 Ready
Future-proofed for next-generation eCTD 4.0 data model with structured content planning, metadata management, and authority submission capabilities.
AmpleLogic RIMS vs. Competitors
How AmpleLogic outperforms legacy RIMS and point solutions
| Capability | AmpleLogic RIMS | Competitors |
|---|---|---|
| Architecture | Low-Code / No-Code on unified aPaaS | Rigid architecture, heavy customization needed |
| Data Approach | Unified master data repository | Fragmented data across modules and silos |
| Lifecycle Coverage | End-to-end (Planning to Post-Approval Tracking) | Often requires separate add-on modules |
| Integration | Seamless (QMS, DMS, ERP, Safety, LIMS) | Standalone, high integration cost and effort |
| Automation & AI | AI-powered alerts, predictive analytics, auto-reports | Limited automation, static manual workflows |
| Total Cost of Ownership | Lower TCO, rapid deployment, pay-as-you-grow | High licensing fees and ongoing maintenance costs |
| Deployment Speed | Weeks with pre-validated templates | Months to years of implementation |
| eCTD 4.0 Readiness | Future-proofed with structured data model | Roadmap-dependent, uncertain timeline |
RIMS Enterprise Integrations
Ten integration modules connecting regulatory, quality, and manufacturing systems
eQMS
Bidirectional change control, CAPA, and deviation linkage
DMS
Version-controlled regulatory documents and SOPs
LMS
Training records and competency management
LIMS
Analytical data and CoA integration for submissions
eLogbook
GxP logbook data linked to regulatory records
eBMR/MES
Batch record data for regulatory submissions
CVS
Cleaning validation data for regulatory filings
CAPS
Equipment calibration records for compliance
SAP/Oracle
Material master and supply chain data sync
PV
Adverse event data and PSUR/PBRER integration
RIMS ROI: Measurable Regulatory Impact
Quantified impact from AI-assisted regulatory lifecycle management
Submission Preparation
50-60% fasterManual Process
8-12 weeks
With AmpleLogic RIMS
3-5 weeks
Regulatory Data Entry
75% reductionManual Process
40+ hours/week
With AmpleLogic RIMS
8-10 hours/week
Deadline Compliance
Near-zero missed deadlinesManual Process
85-90% on-time
With AmpleLogic RIMS
99%+ on-time
Query Response Time
70% faster responseManual Process
5-10 business days
With AmpleLogic RIMS
1-3 business days
Audit Preparation
90% time savingsManual Process
2-4 weeks
With AmpleLogic RIMS
Hours (always ready)
Report Generation
95% fasterManual Process
3-5 days per report
With AmpleLogic RIMS
Minutes (auto-generated)
Featured in Gartner Report
AmpleLogic RIMS has been recognized as a Representative Vendor in the Gartner® Market Guide for Life Science Regulatory Information Management Solutions, September 2024 (ID G00769345).
Industries Served by AmpleLogic RIMS
Eight industry verticals leveraging AmpleLogic RIMS for regulatory excellence
Pharmaceuticals
GMP-compliant regulatory software for drug registration, NDA/ANDA submissions, and global market authorization with full lifecycle management
Biotechnology
BLA submissions, biosimilar registrations, and complex biological product lifecycle management
Medical Devices
510(k), PMA, CE marking, EUDAMED/UDI submissions, and device classification management
Gene Therapy
IND/BLA submissions for advanced therapies with specialized regulatory pathway tracking
API Manufacturers
DMF tracker for CEP applications and global API registration across authorities
Food & Beverages
Food additive registrations, novel food applications, and health claim submissions
Cannabis & Tobacco
Specialized regulatory tracking for cannabis licensing and tobacco product submissions
Cosmetics
Cosmetic product notifications, CPNP/FDA registrations, and ingredient compliance tracking
Ready to Transform Your Regulatory Operations?
Replace fragmented spreadsheets and manual tracking with AI-powered, audit-ready regulatory intelligence.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.