Enterprise AI-Powered Solutions for Every Pharma Workflow
Comprehensive suite of validated software solutions for pharmaceutical, biotech, and life sciences organizations.
Low Code aPaaS for Life Sciences — Unified Integration Architecture
AmpleLogic's modules don't operate in silos. Every solution is interconnected — sharing data, triggering workflows, and maintaining a single source of truth across Quality, Manufacturing, and QC Laboratory functions.
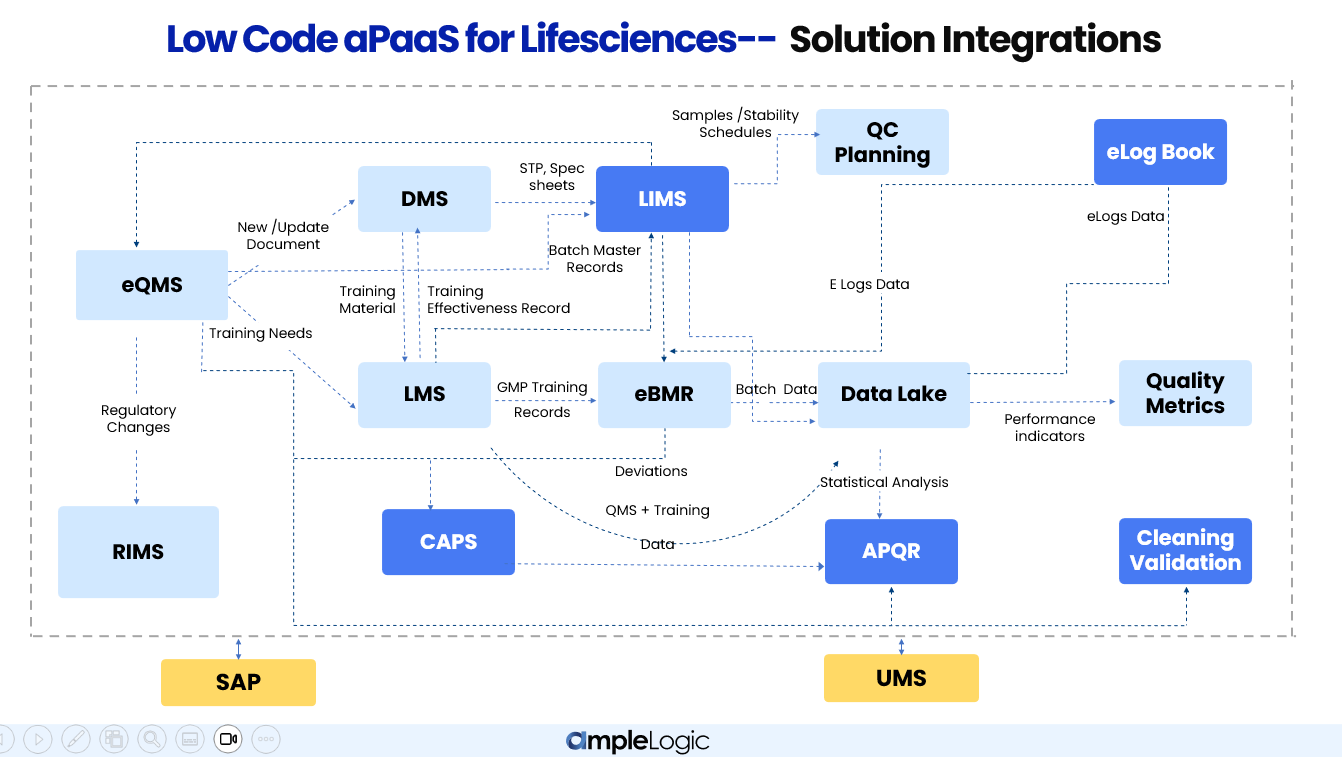
How AmpleLogic modules integrate across Quality, Manufacturing, and QC Lab functions in pharmaceutical companies
Unified Data Lake
All modules feed into a centralized Data Lake, enabling real-time performance indicators, quality metrics, and cross-functional analytics without manual data consolidation.
Automated Workflow Cascades
A single event — such as a deviation in manufacturing — automatically triggers CAPA in eQMS, document revision in DMS, retraining in LMS, and impact assessment in APQR.
Bi-directional Data Flow
Data flows in both directions between modules. For example, LMS sends training effectiveness records back to eQMS, and LIMS returns analytical results to eBMR for batch release.
How Integrations Work Across Functions
Quality Management
The Quality Management ecosystem ensures every deviation, CAPA, change control, and audit finding is tracked end-to-end. eQMS sits at the center — triggering document workflows in DMS, training assignments in LMS, and feeding statistical data into APQR for annual product quality reviews.
New / Update Document
When a deviation or change control is raised in eQMS, DMS automatically initiates an SOP revision workflow, ensuring documents stay current with every quality event.
Training Needs
New or revised SOPs trigger automatic training assignments in LMS. Training effectiveness records flow back to eQMS to close the quality loop.
Statistical Analysis
Batch data, deviations, CAPA records, and process parameters are aggregated in the Data Lake and fed into APQR for comprehensive annual product quality reviews.
QMS + Training Data
Calibration deviations and preventive maintenance records from CAPS flow into APQR, providing a complete picture of equipment and instrument impact on product quality.
Stay Ahead in Life Sciences
Get the latest product updates, compliance news, and industry insights delivered to your inbox.