
How You Can Automate Batch Release in APQR?
Is your batch release process still a manual task of checklists, paper trails, and follow-up emails? You’re not alone. Releasing a pharmaceutical batch is one

Is your batch release process still a manual task of checklists, paper trails, and follow-up emails? You’re not alone. Releasing a pharmaceutical batch is one

Batch release is one of the most scrutinized stages in pharmaceutical manufacturing. It is a regulated step that requires every manufacturing detail to be documented,

Maintaining constant product quality is of extreme importance in the pharmaceutical sector. A thorough assessment of a product’s quality over the course of the year

One of the pillars of the pharmaceutical industry is the concept of Annual Product Quality Review (APQR), which is conducted on each product to verify

Competition is exceedingly high within the pharmaceutical and biotechnology industry whereby quality standards are thus reprehended. This requires constant supervision and investigation of processes involved

In today’s fast-paced and highly regulated industries, Product Review software has become an indispensable tool. Evolving from basic tracking systems to sophisticated, integrated platforms, these

In today’s competitive business landscape, maintaining high product quality and continuously improving offerings is essential for success. APR (Annual Product Review) software has emerged as

Embarking on an exploration of the essential aspects surrounding Annual Product Quality Review (APQR), this investigation delves into the fundamental questions of What, Why, and

The Annual Product Quality Review (APQR) is a critical process for pharmaceutical companies to evaluate the quality standards of their products, ensure regulatory compliance, and

In the intricate and ever-evolving landscape of modern industry, where the quest for operational excellence is both a strategic imperative and a cornerstone of sustained

In the dynamic world of business, maintaining impeccable product quality stands as an unyielding cornerstone. Whether it’s the allure of a cutting-edge smartphone, the aroma

Embracing innovation is a mandate for businesses to remain competitive and advance with futuristic solutions. Continuous Process Validation (CPV) emerges as a transformative approach, offering

Efficiently managing processes is necessary for achieving business operational excellence. A cornerstone of this pursuit lies not only in adept quality management but also in

Maintaining premium product quality and consistency is pivotal in the life sciences industry to ensure safety and reliability. Manufacturers adhere to stringent regulatory requirements while

In the pharmaceutical realm, upholding stringent standards of quality and regulatory compliance stands as a backbone in guaranteeing the safety and efficacy of medicinal offerings.

In the life sciences industry, precision is necessary for maintaining the highest standards of quality and for regulatory compliance. Continuous Quality Improvement (CQI) stands as

Guaranteeing safety, efficacy, and quality of drugs stands as utmost priority in the realm of pharmaceuticals. Previously, achieving this has leaned heavily on batch testing,

Ensuring the highest standards of quality and compliance is essential in the life sciences sector to guarantee safety and effectiveness of medicinal products. Given the

Product quality assurance is primal in pharmaceutical manufacturing. With regulations evolving and the complexity of products increasing, it’s essential for companies to implement robust quality

Quality assurance is paramount in the realm of pharmaceutical manufacturing. The processes of quality management within this industry are rigorously structured and meticulously regulated to

Effective management of excursions is crucial in pharmaceutical manufacturing to uphold both product quality and regulatory compliance. Integrating technological solutions into Product Quality Review (PQR) workflows offers

In pharmaceutical industry, ensuring quality and consistency of products is a top priority. Achieving this goal requires a commitment to continuous process improvement, a cornerstone
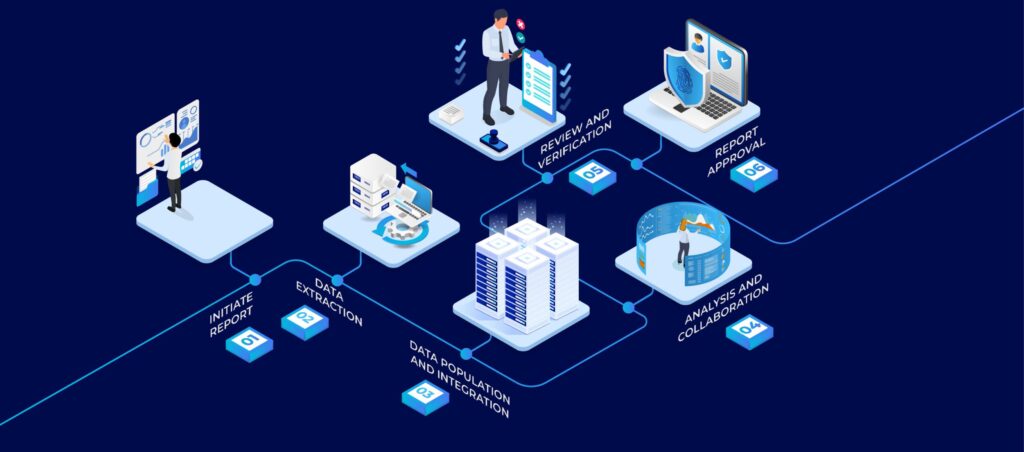
In the dynamic landscape of the Life Sciences industry, ensuring compliance with Good Manufacturing Practices (GMP) is imperative to guarantee the safety, efficacy, and quality
We use cookies to improve your experience on our site. By using our site, you consent to cookies.
Manage your cookie preferences below:
Essential cookies enable basic functions and are necessary for the proper function of the website.

Annual Product Quality Review

Electronic
Logbook System

Laboratory Information Management System

Manufacturing Execution System
Calibration & Preventive Maintenance

Regulatory Information Management System

Learning Management System

Environmental Monitoring System

Electronic Quality Management System

User Access Management System

Document Management System

Quality Control
System

Cleaning Validation
Software

Process Validation
Software

Regulatory Surveillance Management System
Continued Process Verification